Reprinted from the Proceedings of the Second International Conference on Remediation of Chlorinated and Recalcitrant Compounds, May 2000 with an Addendum from ICAM 2000
PIMS - REMEDIATION OF METAL CONTAMINATED WATERS AND SOILS
James L. Conca, Ningping Lu, Gary Parker, Beverly Moore, and
Andrew Adams (LANL, Los Alamos, NM)
Judith Wright and Paula Heller (UFA Ventures, Inc., Richland, WA)
ABSTRACT: Phosphate-Induced Metal Stabilization (PIMS) stabilizes a wide range of metals, especially uranium (U), plutonium (Pu) and lead (Pb), in situ or ex situ, by chemically binding them into new stable phosphate phases and other low-solubility phases that are stable over geologic time. Although some form of apatite is necessary, most apatites do not have the suitable characteristics of no substituted F, high degree of substituted CO32-, low initial trace metal concentrations, extremely poor crystallinity coupled with random nanocrystallites, and high microporosity necessary for optimal performance in the field. A special form of apatite, Apatite II, has been found to be the most reactive, most cost-effective apatite available, having optimal structural and chemical characteristics for metal and radionuclide remediation. This technology has been successfully demonstrated on contaminated soil and groundwater for U, Pb, Pu, Cd, Zn and Cu from mining sites and from DOE sites.
INTRODUCTION
PIMS is a remediation technology that uses a special form of apatite, Apatite II, to clean up metal and radionuclide contaminated soils and groundwater, and works especially well for Pb, U, Pu, Zn, Cd, Cu and other metals. It is simple and straightforward, requiring no sophisticated machinery, and can be emplaced as a down-gradient permeable reactive barrier, mixed into contaminated soil or waste, used as a disposal liner, or emplaced by other methods (Figure 1). Mobilization and removal of metals from the subsurface has not been very successful because of the various intermediate solubilities and sorption properties that each metal and suite of metals exhibits under most environmental conditions. Alternately, metals can be stabilized in place. Many reactive materials have been proposed for this purpose, but Apatite II is particularly effective for U and Pu with their complex redox behavior, and for non-redox-sensitive metals for which no adequate, cost-effective alternatives exist, e.g., Pb, Cd and Zn. The metals sequestered in apatites form new minerals that have great durability and leach resistance significantly exceeding other chemically stabilized waste forms, because the apatite structure is stable over a wide range of conditions, e.g., pH 2 to 12, up to 1000°C, and under disruptions such as earthquakes, ground subsidence or human intrusion, for geologically long time periods, i.e., millions of years (Wright, 1990; Wright et al., 1987). Reactions are fast, occurring in minutes. The bioavailability of ingested metal-apatite is also reduced or eliminated, making animal and human intrusion less dangerous should the material be ingested, and making bioremediation more effective in mixed waste environments. Some form of apatite is necessary for this technology, especially for instigating the heterogeneous nucleation of metal-apatite phases necessary for remediation under environmental conditions (Lower et al., 1998). The excellent stabilization efficiency comes from the extremely low solubility products of the resultant metal-apatites, e.g., for U-phosphate (autunite) Ksp = 10-49, and for Pb-apatite (pyromorphite) Ksp = 10-80. There are several apatite sources with widely varying reactivities and properties, but most are not appropriate for metal remediation. For best results, the apatite should: 1) have as much carbonate ion substituted as possible, 2) have no fluorine substitution, 3) have no trace metals initially in the structure, 4) be poorly crystalline or even amorphous, and 5) have high internal porosity (Conca, 1997). For these reasons, traditional phosphate ores and cow bone, charred or not, are not as effective as Apatite II. Apatite II is also the best material available for non-specific metal adsorption, is an excellent buffer for neutralizing acidity through PO43-, OH-, and substituted CO32-, and exerts control on the chemical activities of other species leading to the precipitation of oxide-, hydroxide- and carbonate metal phases. Non-apatite phosphate and mixtures of precursor constituents will not perform as well, if at all, under most environmental conditions.
FIGURE 1. Some emplacement technologies for Apatite II in remediating heavy metals in contaminated soils, sediments and groundwater.
The ultimate driving force for the robust performance of reactive phosphate with respect to metals is the extreme stability of these metal-phosphate phases, some of which are listed below. These metal phosphates are twenty to seventy orders of magnitude more insoluble than quartz. Combined with this stability, the rapid kinetics of the metal-phosphate precipitation ensures immobilization of the metals in the face of most possible transport mechanisms.
Mineral Phase Solubility Product (log Ksp) Mineral Phase Solubility Product (log Ksp) Pb5(PO4)3(OH,Cl) -76.5 Am(PO4) -24.8 Ca(UO2)2(PO4)2 • 10H2O -49.0 Pu(PO4) -24.4 Sr5(PO4)3(OH) -51.3 UO2HPO4 -10.7 Zn3(PO4)2 -35.3 Quartz (SiO2) -4.0 Cd3(PO4)2 -32.6 Salt(NaCl) 0.0
URANIUM REMEDIATION AT OAK RIDGE AND LOS ALAMOS
Various reactive materials were tested for the removal of dissolved uranium (U) from DOE Y-12 facility NT-1 groundwater at Oak Ridge, Tennessee (Bostick et al., 1999) including 3 different types of bone char, a mixed valent oxide iron filings, granular activated charcoal (GAC), phosphate rock, Nucon Nusorb, and Mersorb-3. The difficulty at this site is that the groundwater can have high total dissolved solids, especially nitrate ion, and contains elevated levels of many regulated metals including U, Cd, and Pb. Apatite II was shown to be almost ten times as effective as bone char for removing U, and many orders of magnitude more effective than the other materials including zeolite, activated charcoal, peat moss, zero valent iron and other forms of reduced iron, even in the presence of high nitrate concentrations. Apatite II was also much more cost-effective. Some of the results are shown in Figure 2. X-ray diffraction after batch tests showed meta-autunite crystallized on the surfaces of the Apatite II.
Apatite II is also being used as part of a Containerized Vat Leaching process (CVT) at a depleted-uranium firing range. Soil contaminated with U, is leached with bicarbonate solution to remove the U. The U is then removed from solution using Apatite II instead of exchange resins. The process
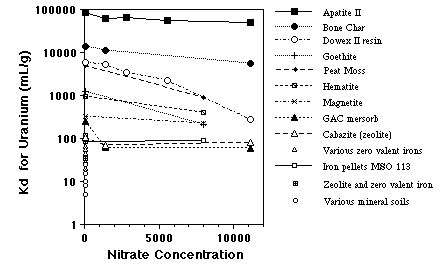
FIGURE 2. Sorption results for various reactive media in U-contaminated groundwaters from Oak Ridge. Apatite II performed better than all other materials even under high nitrate concentrations.
is relatively inexpensive and simple. Figure 3 shows the treatment pathway of contaminated soil. Soil, mixed with a small amount of Na2O2 to oxidize any reduced U to U(VI), is heaped into containers, then infiltrated from below with bicarbonate solution that goes into a settling- acidification basin and then into sorbent canisters filled with Apatite II. The solution is then recarbonated. The cleaned soil is sampled, placed back on the ground, or used for other purposes, and the concentrated U-product is either disposed of, or processed in a mill. Apatite II in this case is coarse-grained. Figure 4 shows the U concentration going from the effluent exiting the leaching step (37 ppm U) to the effluent exiting the Apatite II (less than 0.01 ppm). The volume reduction in contaminated materials is on the order of 1000 times. TEM images of the Apatite II after treatment showed U covering all available surfaces of the Apatite II. A phase determination has not yet been made, but autunite is expected.
Pu RETARDATION AT WIPP
PIMS was investigated as a possible backfill component at the Waste Isolation Pilot Plant (WIPP) in Carlsbad, NM, to prevent Pu(VI) migration from transuranic waste. Experiments using Pu(VI) in WIPP Brine A infiltrating through various backfill combinations showed that a layered backfill of apatite/MgO/montmorillonite performed well, and performed better layered rather than mixed. Placing the apatite as the first layer increased performance over 100 fold. Figure 5 shows results from one of the layered backfill column experiments with a 50% (apatite-25% MgO-25%) montmorillonite barrier. Initial infiltration concentration of 570 CPM/ml dropped to 10 CPM/ml after moving through the barrier. Studies are underway to determine the phase and speciation of the Pu in the barrier and in the effluent.
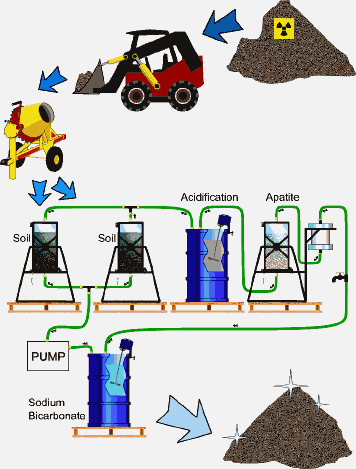
FIGURE 3. PIMS used with Containerized Vat Leaching to remediate U-contaminated soil at a firing range.
Pb, Cd, Zn AND Cu AT BUNKER HILL AND SUCCESS MINES
The metal-stabilization potential of reactive phosphates from different sources was investigated along with other materials for remediation of contaminated soil and groundwater from the Bunker Hill Site and the nearby Success Mine Site in northern Idaho (Chen et al., 1997; Wright et al., 1995; Conca, 1997). Soils were contaminated with 1000-4000 mg kg-1 (ppm) of Pb, Zn, Cd and Cu, and ground-waters were contaminated with Zn, Pb, Cd and Cu up to concentrations of 250 ppm, 10ppm, 1 ppm and 20ppm, respectively. Apatite II stabilized almost 20% of its weight in Pb, and about 5% of its weight in Zn and Cd. Apatite II completely outperformed other additive materials such as zeolites (clinoptilolite and chabazite), compost, various polymers, iron filings, and other
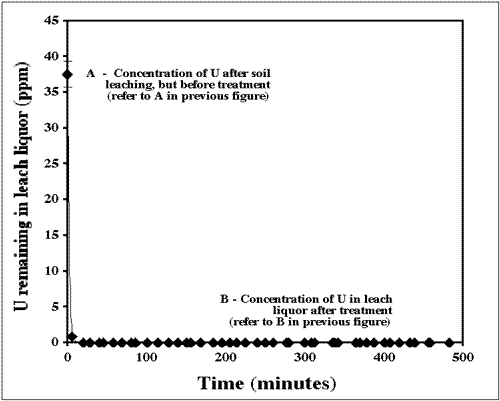
FIGURE 4. Uranium in the effluent exiting the leaching step (A) and exiting the Apatite (B) in Figure 3
apatites, for Pb, Cd, and Zn. The bioavailability of the metals from the contaminated soil was also greatly reduced even when the metal was not in an apatite phase. Pb precipitated as lead-pyromorphites while Zn and Cd both sorbed onto particles and precipitated as hopeite, zincite hydrocerrusite, otavite and other phases. Results showed that the leachates from untreated soils were in the hundreds to thousands of ppm range for Pb and Zn, and hundreds of ppm for Cd, well in excess of regulatory Maximum Contamination Levels for drinking water. However, leachates from PIMS-treated soils showed Pb, Zn and Cd below the detection limits of 1 ppb and 5 ppb using Inductively-Coupled Plasma Mass Spectrometry (ICP/MS) and Potentiometric Stripping (Chen et al., 1997; Wright et al., 1995; Conca, 1997). Performance was successfully predicted using MINTEQ-A2, a thermodynamic speciation model (Moody and Wright, 1995), was substantiated by XRD results on the contaminated soils after PIMS treatment (Wright et al., 1995) and was also consistent with results by others researchers (Lower et al., 1998; Ma et al., 1993).
In preparation for emplacement of a permeable reactive barrier to treat Zn, Cd and Pb in seep waters from the Success Mine tailings pile, a feasibility study was performed on various materials, including different sources of apatite [three Apatite II formulations (Apatite WE, PR, and AP) and cow-bone apatite (Apatite CB)], iron-filings, compost/woodchip/gravel mixture, two zeolites and a polymer used in remediation of mine wastes, and activated charcoal. The Apatite II performed the best with respect to all metals. The results for Zn are shown in Figure 6 which plots contaminant concentration in the effluent normalized to the influent, C/C0, versus the volume of water passing through the column normalized to the weight of barrier material. Similar results occurred for Cd (Conca, 1997).
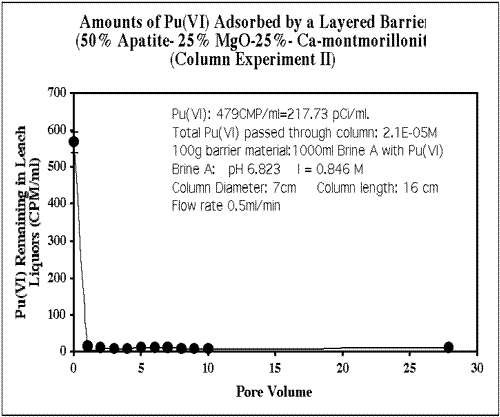
FIGURE 5. Pu(VI) retardation by a layered Apatite/MgO/Montmorillonite engineered barrier at WIPP
Batch leach tests were performed to provide a relative effect of Apatite II on the bioavailability of Pb from ingested contaminated soils in water foul, a severe problem in this area. Contaminated soil was collected and treated with 1%, 10% and 50% Apatite II by weight. These were run in batch tests adjusted to pH 2.4 with HCl. The substrate to water ratio was the standard 1:10 and mixtures were shaken for 24 hours. The solutions were then filtered through 0.2 mm filters and analyzed for Pb and Zn. The results are given below. Ongoing toxicity tests also show significant decreases in Pb uptake into organs and bone with the addition of Apatite II to contaminated soil (unpublished proprietary reports).
Soil Pb Zn pH untreated soil 24,000 ppb 2,800 ppb 4.28 1% Apatite II 174 ppb 140 ppb 6.07 10% Apatite II 104 ppb 50 ppb 6.87 50% Apatite II 240 ppb 50 ppb 7.02 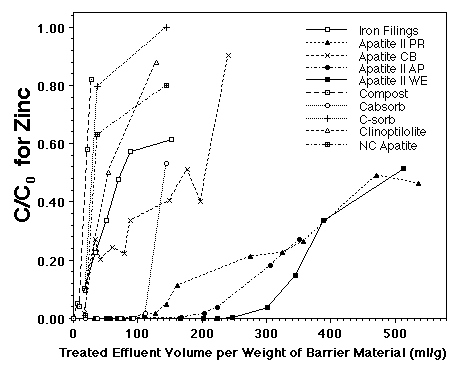
FIGURE 6. Breakthrough of dissolved Zn through various permeable reactive barriers using contaminated groundwater from the Success Mine Site. Apatite II performed best.
ADDENDUM - Apatite Reactivity and Structure
The special reactivity of Apatite II comes from the unusual structure of the solid. Most of the material is amorphous hydroxy calcium phosphate with respect to both XRD and to TEM. However, there are random nanocrystals of well-crystallized apatite (Figure 7). Also, there is substantial carbonate substitution which makes the structure less stable and more reactive (Wright et al., 1990). However, once metals are in the apatite structure, it is much more stable than the initial Apatite II. Because of this arrangement, the amorphous structure is relatively reactive and provides a slight excess of phosphate ion to solution, locally exceeding the solubility limit of the metal-phosphate phase and resulting in precipitation. At the same time, the nanocrystals provide the apatite structure for nucleating the precipitates. Other apatites, such as phosphate rock, bone char, or reagent grade tricalcium phosphate, are crystalline (Figures 8 and 9), have little or no carbonate substitution, have substantial fluorine substitution, have significant trace metal substitution (Sr, Ba, etc.), or have less microporosity, all of which reduce the reactivity and make them less optimal for metal stabilization. An example of the efficiency of Apatite II is shown in Figure 10, where uranium-contaminated leachate has been remediated using Apatite II in a reactive barrier flow-through situation.
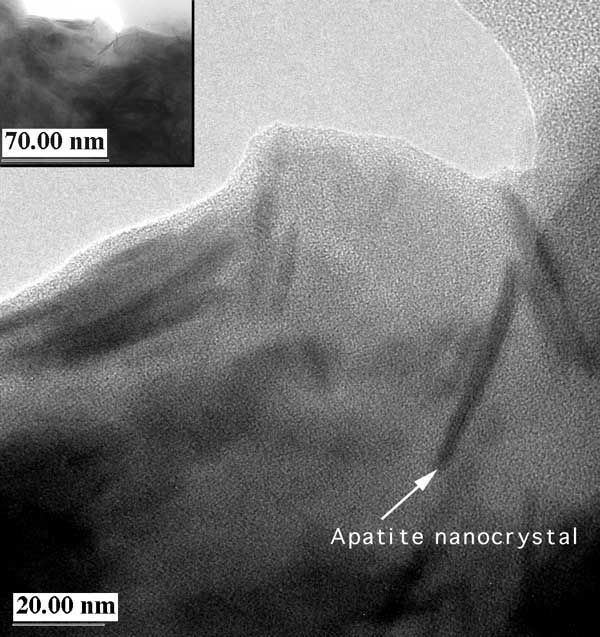
Figure 7. HR-TEM image of Apatite II showing general amorphous nature with random nanocrystal inclusions of crystalline apatite (low-resolution image is inserted in upper left corner).

Figure 8. HR-TEM image of mineral apatite (NC phosphate rock) showing the crystalline nature of the apatite.

Figure 9. HR-TEM image of bone char showing the crystalline nature of the apatite.

Figure 10. HR-TEM image of Apatite II (same as in Figure 7) after treatment of leachate from uranium-contaminated soil during a soil washing operation. In this case, Apatite II was chosen over exchange resins to remove uranium from the leachate because of its efficiency and low cost. The uranium is completely covering the surface of the Apatite II.
CONCLUSIONS
PIMS, using Apatite II is a significant new remediation technology providing the following benefits.
- Apatite II stabilizes most heavy metals and radionuclides for geologically long time periods.
- Many metal and radionuclide contaminants are precipitated as mineral phases in which the metals are not bioavailable.
- Apatite II is the best non-specific surface sorbent for species that do not precipitate as a separate phase.
- Apatite II is the most cost-effective reactive media for most metals and radionuclides, e.g., immobilizes up to 20% of its weight in Pb, U, Pu and perhaps other metals.
- Apatite II can be mixed into contaminated soil or waste, emplaced as a down-gradient permeable reactive barrier, or used as a disposal liner.
- Apatite II can be combined with most other reactive media and other remediation technologies, especially for mixed wastes, e.g., grout, bentonite, zero valent iron, vapor stripping, bioremediation, etc.
Phosphates other than apatites are not effective for the purposes of metal and radionuclide remediation, and apatites other than Apatite II are much less effective than Apatite II for this purpose.
REFERENCES
Bostick, W. D., R. J. Jarabek, D. A. Bostick, and J. Conca .1999. "Phosphate-Induced Metal Stabilization: Use of Apatite and Bone Char for the Removal of Soluble Radionuclides in Authentic and Simulated DOE Groundwaters." Advances in Environmental Research. 3: 488-498.
Chen, X.-B., J. V. Wright, J. L. Conca, and L. M. Peurrung. 1997. "Evaluation of Heavy Metal Remediation Using Mineral Apatite." Water, Air and Soil Pollution 98: 57-78.
Conca, J. L. 1997. Phosphate-Induced Metal Stabilization (PIMS). Final Report to the U. S. Environmental Protection Agency #68D60023, Res. Triangle Park, NC.
Lower, S. K., P. A. Maurice, S. J. Traina, and E. H. Carlson. 1998. "Aqueous Pb sorption by hydroxylapatite: Applications of atomic force microscopy to dissolution, nucleation and growth studies." Amer. Min. 83: 147-158.
Ma, Q. Y., S. J. Traina, and T. J. Logan. 1993. "In Situ Lead Immobilization by Apatite." Env. Sci. Tech. 27: 1803-1810.
Moody, T.E., and J. Wright. 1995. Adsorption Isotherms: North Carolina Apatite Induced Precipitation of Lead, Zinc, Manganese and Cadmium from Bunker Hill 4000 Soil, Technical Report BHI-00197, Bechtel Hanford, Richland, WA.
Wright, J. V., L. M. Peurrung, T. E. Moody, J. L. Conca, X. Chen, P. P. Didzerekis, and E. Wyse. 1995. In Situ Immobilization of Heavy metals in Apatite Mineral Formulations, Technical Report to the Strategic Environmental Research and Development Program, Department of Defense, PNL, Richland, WA.
Wright, J. 1990. "Conodont apatite: structure and geochemistry." In Joseph Carter (Ed.), Metazoan Biomineralization: Patterns, Processes and Evolutionary Trends. pp. 445-459, Van Nostrand Reinhold, New York.
Wright, Judith, James L. Conca, John Repetski, and James Clark (1990) "Microgeochemistry of some Lower Ordovician Cordylodans from Jilin, China." In Willi Ziegler (ed.) 1st International Senckenberg Conference and 5th European Conodont Symposium (ECOS V) Contributions III, Courier Forschungsinstitut Senckenberg, Frankfurt, Germany, vol. 118, p. 307-332.
Wright, J., H. Schrader, and W. T. Holser. 1987. "Paleoredox Variations in Ancient Oceans Recorded by Rare Earth Elements in Fossil Apatite." Geoch. Cosmoch. Acta 51: 631-644.